Study of the preparation of soap prepared from oils different
Muhannad Abdel Salam Osman1
Teacher at Nineveh Education, Mosul, Iraq.
Email: mohothmn@gmail.com
HNSJ, 2024, 5(5); https://doi.org/10.53796/hnsj55/14
Published at 01/05/2024 Accepted at 05/04/2024
Abstract
Oils and fats are important to the soap manufacturing industry because they contain glycerides and non-glycerides, the latter of which is considered a pest. Since oils and fats are primarily composed of fatty acids, choosing the appropriate oils and fats is of great importance to the creation of soap. Oil and fat that are appropriate for this method include coconut oil, palm oil, and palm kernel oil.
“The percentage of unsaturated fatty acids in the mixture has an effect on the soap’s energy, capacity for cleaning, hardness, and scent”.
The final properties, including the lathering effect and the moisturizing effect, are primarily derived from the additives or fillers used.
عنوان البحث
دراسة تحضير الصابون من الزيوت المختلفة
مهند عبد السلام عثمان1
1 مدرس بتربية نينوى، الموصل،العراق.
البريد الإلكتروني : mohothmn@gmail.com
HNSJ, 2024, 5(5); https://doi.org/10.53796/hnsj55/14
تاريخ النشر: 01/05/2024م تاريخ القبول: 05/04/2024م
المستخلص
تُعدّ الزيوت والدهون عنصرًا أساسيًا في صناعة الصابون لاحتوائها على الجلسريدات وغير الجلسريدات، والتي تُعتبر الأخيرة منها ضارة. ولأن الزيوت والدهون تتكون أساسًا من الأحماض الدهنية، فإن اختيار الزيوت والدهون المناسبة يُعدّ بالغ الأهمية في صناعة الصابون. ومن الزيوت والدهون المناسبة لهذه الطريقة: زيت جوز الهند، وزيت النخيل، وزيت نواة النخيل.
تؤثر نسبة الأحماض الدهنية غير المشبعة في الخليط على طاقة الصابون، وقدرته على التنظيف، وصلابته، ورائحته.
أما الخصائص النهائية، بما في ذلك رغوة الصابون وترطيبه، فتُستمد أساسًا من الإضافات أو المواد المالئة المستخدمة.
“A brief history of the soap industry”:
Traditionally, people have incorporated herbs into their baths and attributed the promotion of health to them. For instance, Cleopatra of Egypt bathed with scented oils, honey, and mare’s milk. According to historical studies, soap was used 5,000 years ago in ancient Egypt and Babylon. Soap was made by combining alkali plant ashes with animal lipids (1).
In the first century AD, people are said to have used water and wood ashes to treat discomfort brought on by oil or grease. Pliny claims that Germanic people used a soap made of wood ash and tallow to brighten their hair. Although the Phoenicians learned about Makange soap about 600 BC, the Babylonians are believed to have utilized it circa 2800 BC. These early allusions had an impact on the development of soap.
“Since ancient civilizations, the fundamental process of producing soap has remained unchanged. As a result, both old practices and modern knowledge are combined with carefully selected ingredients that are combined and stirred at the proper temperature and duration”. It could lead to experimenting with the finest soaps (2).
Animal fats, such as those derived from deer, are the primary components of soap creation. After the French invented the method of manufacturing soap with olive oil, this technology quickly became popular in the England. Scientists Carl Scher and Scher had an unfortunate discovery of glycerin while heating oil and adding lead oxide to it (3).
In the 14th century, chemists participated in the enhancement of the quality of soap by understanding the properties of the fats and oils involved in its manufacturing. Coincidentally, in 1823 AD, Carl and Shell, the scientist Chevrol, discovered that simple fats break down first to make acids rather than reacting instantaneously with alkalis to form soap. Glycerin and fatty when chemist Nicholas LeBlanc discovered a method for producing soap, the soap business underwent a revolution. Regular salt or sodium carbonate soda (4).
Necessary raw materials for the soap industry:
The common practice of using sodium hydroxide has adversely affected the soap-making process. Soap is made by combining acid with alkali (sodium hydroxide solution). Glycerin, soap, and water. Saponification is the formal term for the procedure. “Almost any fat or oil can be converted into soap by combining it with water. Triglycerides, which are derived from the esterification of glycerol with three long-chain carboxylic acids (L-fats), are the most simple form of animal and vegetable fat (5)”.
“When triglycerides are combined with a water-based mixture that is basic, such as potassium hydroxide or sodium hydroxide, they are cleaved. This produces soap that contains glycerin and unbound fatty acids”. The salts of fatty acids are singular in composition. Oils like the olives, rapes, marbles, and coconuts are involved in the production of soap alongside other substances like milk, avos, and titanium dioxide (6). Soap is composed of chemical hydroxides combined with fats derived from animal and vegetable sources (7, 8).
Animal Lipids:
One of the most essential components of soap creation is butter. Around 2550 BC, the Romans employed wood ashes and animal fats to create soap. “Animal fat soap has a higher concentration of over 16 or 18 fatty acids. It’s typically more time-consuming to create than butter soap (9)”.
1-3 “Using animal fats as a raw source to make soap”:
The manufacturing of soap was one of the most incredible events of Rome, and may have been accidental. Folklore stories that the Romans formerly ate animals as a form of payment to the gods at the top of Sapo near Rome. After the ceremonies, the ashes and animal fat were placed in the fireplaces. When it rains, Roman women would bring their clothing to the riverbanks and find a white, porous substance that would float in the water, this had come from the fat and ash. The women would soak their clothing in the mixture, then scrub it with rocks. The oily substance produced bubbles when in contact with the fabric, this washes away the rest of the fabric. Eventually, people began to derive soap from animal fats (10).
Acids in Fats:
The two most significant components of lipids are fatty acids (FAS) and free fatty acids (FFAS), these are non-flammable aliphatic monocarboxylic acid molecules that can also be utilized as building blocks for soap.
“Fatty acids are common in nature, and their physical properties have been studied in order to determine the most effective and most specific ways to utilize them” (11).
However, to this point, there is no information regarding the detection or diagnosis of free fatty acids using GC-MS (free fatty acid). However, various methods have been attempted to calculate the quantity of metals, residual solvents, moisture, volatile components, natural oils, and total fatty acids in detergents (12, 13).
Initially, free fatty acids must be acquired. These are achieved by releasing fatty substances that are volatile. In this approach, water is employed as a catalyst to break down triglycerides (14).
The quantity of fatty acids produced has an effect on the quality of the soap. Saturated free fatty acids contribute to the soap’s solid properties and to the dense, light layer. Conversely, unsaturated fatty acids that are released have the capacity to condition, nourish, and moisturize (15). Additionally, free bioactive fatty acids have a significant impact on the quality of soap, and their presence is essential to the effectiveness of detergents.
Soaps that contain free fatty acids as a part of their composition are particularly beneficial because they have a higher concentration of emollients and produce a more rich lather, both of these properties are significant (16, 17).
Because of the complexity and difficulty in studying the composition of soap, the majority of the fatty acids that are derived are highly polar, non-intuitive molecules. “As a result, derivatization is essential to increase the thermal stability of dangerous substances. This can be accomplished by reducing the polarity of the analyte, which will lead to an improvement in its properties regarding chromatography in the gas phase” (18).
Oil types used to make soap:
Although the basic principles of soap making are identical worldwide, the methods vary depending on the intended purpose and the type of oils used (animal or vegetable). Below is a list of oils that are produced as byproducts in the soap-making process.
Shea Nut Oil:
“As part of the ongoing EU-funded INCO study on shea butter, Ben-Gurion University in Israel analyzed the chemical composition of shea butter that was derived from the nut samples from four different African countries (Uganda, Nigeria, Burkina Faso, and Mali) (19). The analysis of fatty acids (Table 1) showed that the composition of shea butter was different in Africa than it was elsewhere”.
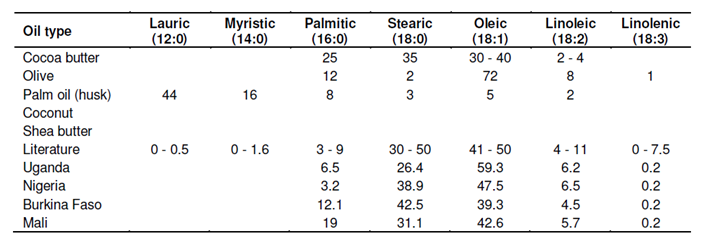
Table (1): Fatty acid studies indicates that shea oils differ significantly across Africa.
The percentage of oleic acid was 59% in the sample from Uganda, 47% in Nigeria, only 39% in Burkina Faso, and 42% in Mali. Solid, fatty shea butter, also known as stearin, is crucial to the manufacturing of soaps and detergents (20).
Shea butter is primarily composed of triglycerides, with the second and third positions being occupied by oleic acid, and the first position being occupied by a fatty acid that is already in a state of saturation (21).
Oil from katropha seeds:
Because the oil in the fruit of the Jatropha curcas is viscous and contains a lot of palmitic acid, it can produce soft, long-lasting soap with the most basic manufacturing process (23), thus it can be utilized in the production of soap (22).
Castor seed oil:
Castor oil has a higher concentration of ricinoleic acid (around 90%) than other vegetable oils, this differentiates it from the other commercial oils. Castor oil also contains a chemical named ricinoleic acid that has the formula CHCHCHOHCH=CH(H)COOH. Additionally, castor oil contains dihydroxystearic acid, palmitic acid, and oleic acid (24).
Because of its high concentration of hydroxy acids, castor oil is highly malleable, has a low solubility in hexane, and is highly soluble in alcohol. These hydroxy acids have various attributes that allow for their conversion to useful substances (25).
Neem seed oil:
“Neem seed oil is composed primarily of triglycerides, oleic acid, stearic acid, linoleic acid, and palmitic acid”. The dark yellow neem oil is known for its exceptional quality, constituting approximately 40% of neem oil (26). “Neem oil has been a common ingredient in Indian soap making for at least half a century (27)”.
Sesame seed oil:
The seed is referred to as the “queen of seeds that contain oil,” because of its exceptional attributes and large volume of oil.
Other than being employed in scents and cosmetics (28), the heated oil extract is commonly utilized to create soap (29).
“The physical and chemical properties of the oil derived from Nigerian sesame are listed in Table (2) (30). White oil has a composition of 48 percent oil and a value for saponification of 189, while red oil has a composition of 50 percent oil and a value of saponification of 191”.

Virgin Coconut Oil:
Virgin coconut oil contains a larger amount of lauric acid (46%) and a stronger saponification effect than ordinary coconut oil. It works well as a fundamental ingredient in natural liquid soap and is appropriate for wet skin (31).
To create soap with quality, it’s crucial to research the method of combining palm oil with other seeds that have a high capacity for saponification. To the best of the authors’ knowledge, there have been no documented instances of the utilization of rubber tree oil or palm kernel oil in the production of soap (32). The literature describes the substances necessary for the production of soap (33).
Factors affecting the soap industry:
According to Classic Belle (31), soap derived from ammonia (NH₄OH) is more soluble in water than the same amount of soap derived from potassium hydroxide (KOH). The soap industry has undergone significant changes as a result of the consistent evolution of superior soap quality.
The utilization of new substances and augmentations to these materials has been studied. The characteristics of oils, the supply of raw materials, and the cost efficiency of production are all considered in the soap industry (34).
The soaping process:
Soap is produced by combining chemicals associated with soap with animal and vegetable fat as well as temperature elevation, this is beneficial for manufacturing (35). Since soap is a mineral that contains fatty acids and surfactants, it can be employed to clean objects (36).
“Ainley et al. believe that soap is comprised of sodium salts of fatty acids like lauric acid, oleic acid, myristic acid, stearic acid, and palmitic acid (37)”.
Carl notes that many manufacturers have a mixture of 20% of the fatty acids found in coconut and 80% of the fatty acids found in tallow in their products. Describe how to utilize palm oil in place of lard or coconut oil (38).
“On the other hand, Egyptian toilet soap is composed of tallow or stearic acid derived from the shells of palm trees in conjunction with 15% of the tree’s oil (39)”. “Tan et al. believe that carotenoids are the cause of the orange-red color in raw oil of palms (40)”.
How the saponification process works:
Soap’s diverse array of uses includes cleaning, bathing, and pharmaceuticals. The cleaning ability of soap is derived from the anions on its hydrocarbon chains, which are bonded to the carboxylic acid groups of fats (34).
Saponification is the procedure in the production of soap that involves the degradation of stearic acid in an alkaline solution, this produces long-chain hydrocarbons with a variety of ends and groups that can attach to metal ions, typically sodium or potassium ions.
The process of saponification is depicted in Figure 1.
How to Create Soap:
Figure (1) The process of saponification. Additionally: how is soap created?
These molecules that have long chains of carbon are typically called “soap.”
“The hydrocarbon chain’s final end (hydrophobic end) is nonpolar and can be dissolved in solvents that are not polar, such as oil. However, its metal ion component (hydrophilic component) is polar and can be imbued with polar solvents, such as water”.
Soap as a Cleaning Agent Nevertheless:
Since grease is unable to polarize, water will not have a significant effect on the removal of simple dirt. When soap is employed to Remove grease or dirt, the nonpolar parts of the molecule are attracted to the dirt, which forms a micellar layer, following the concept of “like dissolves like” (42).
“When water is added to the soap, the polar components of the molecule’s molecules immediately associate with the polar components of water”.
“The mixture that is derived from this is referred to as an emulsion, because the soap is involved in the association of water with dirt”.
“The emulsion can then be flooded with water to Remove the grease from the top (41)”.
Chemical properties of soap:
The chemical properties of soap are affected by factors like the type of oil, the concentration and purity of the lye, the degree of saponification during use, and the shelf life of the product.
“Other attributes include the moisture content, the total amount of fatty acids (TFM), the pH value, the amount of free lye, the percentage of unsaponifiables, the foaming effect of stabilizers, and the density of the soap. The physical and chemical properties of the soap were also considered (43)”.
Research problem:
The difficulty of this endeavor is in understanding and investigating the different manufacturing processes of oils and fats.
After the French invented the process of manufacturing soap with olive oil, the industry’s expansion became more popular. This was followed by England. Scientists Charles Shear and Shear had the opportunity to discover glycerin when they blended olives with lead dioxide. In the 14th century, chemists had a significant impact on the quality of soap by studying the properties of the oils and fats involved in the manufacturing of soap.
research importance:
This study’s significance stems from the oils used in the soap business, one of the major sectors.
research aims:
- Learn a little bit about the soap industry’s past.
- The oils used to make soap.
search limits:
This chapter discusses animal and vegetable fats, these are important to the soap industry regarding raw materials.
Define terms:
“Vegetable and animal fats are primarily composed of triglycerides, which are derived from the esterification of three molecules of glycerol with three different carboxylic acids (long-chain fatty acids). When triglycerides are combined with basic aqueous chemicals like potassium hydroxide or sodium hydroxide, the fatty acid esters are dissociated. These salts of fatty acids have unique properties that make them stand out”.
The methodology and procedure of research.:
In order to provide a theoretical explanation for the observed variables, this research followed the hypothetical-deductive approach.
A collection of hypotheses is provided by preceding theories and the investigation, which is based on prior research.
After that, test these theories to get a set of findings that are applicable to the whole research population.
Theoretical framework and previous studies:
One of the most significant manufacturing industries that countries devote enormous resources to is the soap industry. This industry is primarily reliant on the raw materials utilized. These raw materials include various oils and fats, which will be discussed in this chapter.
Previous attempts at this endeavor: Medullary acetate is derived from the physical deterioration of acyl chloride. “Medullary acetate is primarily composed of free fatty acids (82.2%), it has a low concentration of neutral fatty acids (11.9%). The remaining components are soluble in water and have impurities (2.1 percent) in addition to water (3.8 percent)”.
The outcomes demonstrated that bleaching enhanced the color of the soap and increased the volume of butter and palm kernel oil in the soap. We created eight different samples of soap (8-1), each having a different composition of derelictly-palmated oil, butter, and Medullary acetate.
The outcomes demonstrated that the water content of the soap samples… Since the total fat content of the samples (7, 2) and (8) was greater than the control sample (1), palm oil was incorporated into soap (44).
Using the waste from the surrounding area as the starting material and a 20:80 ratio of gum to oil (palm), a liquid detergent (palm oil) was created.
…Duncan’s multiple comparison approach was employed to determine. The oil was decolored in order to eliminate its dark color, it was then purges with activated carbon and compressed air in a laboratory.
… The response of saponification (130.5) and the creation of local and experimental-based potassium oxide (126.3) were recognized.
… The expert’s evaluation revealed that soap derived from hydroxide in the local area had a superior effectiveness (45) than the laboratory-produced hydroxide.
This research utilized a unique methodology that was accurate, dependable, and efficient in determining the exact composition of free fatty acids in soap and detergent samples.
This investigation sought to assess the effectiveness of acylation as a means of recognizing free fatty acids in soap samples using either mass spectrometry or gas chromatography. A detector and ammonia were employed as chemical initiators. Chloroform was the solvent and hexamethyldisilazine (HMDS) was the chemical catalyst for a practical and cost-effective process of esterification. Gas chromatography-mass spectrometry (GC-MS) analysis was conducted on a capillary column (5-DB). The linearity, consistency, LOD, and LOQ of the method were documented.
The method was evaluated with a coefficient of determination that was greater than 0.999, this demonstrated a strong association between the concentration and the slope across the entire range of concentrations (2–0.005 mg/ml).
The regression analysis produced LODs of 2.5 pg/ml and LOQs of 8 pg/ml, respectively. Four different types of soap samples with different levels of quality were evaluated using the approved method. The specific amount of free fatty acids in the soaps was from 1.24% to 5.09%. This approach can be employed to determine the percentage composition of free fatty acids in different soap samples (46).
Shea butter is derived from the edible part of the shea tree (Butyrospermum parkii), this part is native to the African savanna. This oil is derived from the fruit’s skins using a cold method, this is then converted into soap with medicinal properties. Chemical analysis demonstrated that the final soap had a composition of 76.0%, 9.0%, 3.41%, 9.0%, 0.00%, 3.7%, and 0.87%.
Key attributes, in order of decreasing importance, were: pH, the total amount of fat, moisture, the stability of the foam, the content of non-soluble matter, the free amount of alkali, and specific gravity. The shea butter’s base is made up of plants and the chemical properties of soap that are beneficial to utilize in medicated soaps and cosmetics that alleviate the skin and scalp. These matters (47).
To gather information that is crucial to completing the task and determine the most effective methods of production, we researched the local soap industry regarding the production of liquid soap.
The results demonstrated that machine-made soaps for consumption in liquid form were more cloudy and sluggish than hand-pouring soaps (48).
We combined two animal oil-based combinations with a vegetable-based one that had the same foaming rate associated with three different mineral oils in a percentage of 3% (1%). We inspected the pH, conductivity, buoyancy, hardness, and size of the final particles of soap.
We contrasted the effects of different types of mineral oil and assessed the results. This investigation described the effects of adding mineral oil to soap samples (36). Another investigation considered the eradication of five different types of soap derived from Nigerian oils: shea butter, tallow, and palm oil.
The samples of soap were then left to dry in air for 21 days, the daily amount of water lost was documented to assess the procedure.
Brinell’s hardness was determined using a homemade handheld device with a weight of 6.585 kgf (64.553 N).
All of the soaps demonstrated a similar pattern of behavior during the curing process, as demonstrated by the one-day moisture content loss curve (41).
The results demonstrated that the hardness of the soaps changed as follows: tallow (HN 1.88) > HN 0.3, shea butter (Palm) > Palm oil (HN 0.25), peanut oil (HN 0.13) > Palm kernel oil (HN 0.15).
A investigation studied the methods of testing for soap at the American Institute of Petroleum Chemists (AOCS).
This is a practical method for calculating and evaluating the amount of oil.
To determine if the raw material is suitable for further processing that would lead to a higher quality product. the quality of the finished soap can be determined and anticipated by testing the peroxide value, the iodine value, and the value of saponification of the raw oil used (49). Virgin coconut oil has a 46% concentration of lauric acid, which causes it to have a more potent saponification than regular coconut oil.
Virgin coconut oil (VCO) is converted into free fatty acids which can be utilized as a raw material for manufacturing soap. VCO is beneficial as a supplement to natural liquid soap.
This research aims to create soap with a higher water solubility using KOH and NH₄OH as alkalis.
This investigation studied the process of saponification between the VCO and alkali in order to determine the quality of natural liquid soap.
This research also sought to determine the effect of the amount of alkali, temperature, and time of mixture on the quality of the soap.
This research utilized response surface methodology. Physical and chemical analyses were conducted on the finished soap.
To enhance the efficiency of the liquid soap, the maximum amount of KOH that could be incorporated into the reaction was 8, which was achieved after 140 minutes of reaction.
The temperature of the reaction was 92 degrees Celsius.
Sample 6 (SNI 1996-4085-06) had the greatest experimental success. The value of critical free fatty acids was 0. SNI 721.0 3532-2016 (31).
This research sought to produce liquid soap using the sheep’s fat from the Tsuna sheep in the Alhangai region. Sheep’s tail fat (TF), the outer skin’s oil (OF), and the inner skin’s oil (IF) were employed as the raw materials for oil creation.
The base of the soap was isolated chemically, while the liquid soap was created using a cold process. The residual oil (TFO) had the greatest concentration of unsaturated fatty acids (41.23% of the total, 22.3% of the total, and 33.65% of the total), and its composition was confirmed by GC-MS (50) as it was the most beneficial for manufacturing liquid soap.
This investigation sought to explore the purposes of residual oils (primarily from private residences). The investigation concerned the production of handmade soap and the phenomenon that the degradation of soap is more significant than the treatment of oil.
The physical and chemical composition of the soaps was confirmed to be different between samples derived from fresh and fried oils.
Significant discrepancies were noted between soaps (p < 0.05).
Soaps were created using freshly harvested olives and also used vegetable oil as a vehicle for frying. A research study on the degradation of soap demonstrated that soap was four times more effective at degrading than oils in wastewater treatment plants.
Based on the literature, the results suggest that soaps derived from vegetable oil fried in the environment are both economically and environmentally viable. Soap manufacturing is a practical strategy for combating oil waste emissions (51).
Clay, also called kaolin, is employed as a component of cosmetics because it can adsorb a variety of substances, including oils and other pollutants.
This investigation sought to explore the effects of kaolin addition and temperature on the properties of the finished solid soap. The saponification reaction was conducted in a reaction vessel that was temperature dependent, with a range of 50-80 degrees Celsius for 10 minutes.
Coconut oil was heated to the intended temperature and then incorporated into different concentrations (10%, 12.5%, 15%, 17.5%, and 20% weight percent) of kaolin.
Later, a 35% solution of sodium hydroxide was incorporated. The mixture’s reaction was stirred at a speed of 250 rpm.
Based on the results of the test, each product from soap that was tested met the requirements of the Indonesian SNI standard (52).
When coconut oil and castor oil were incorporated into a 3:1 ratio, the total fat content (TFM) was maximized at 76.8 percent, with a yield of 89.46 percent.
The water content of the mixed oils (SAP value of 230.4, also an iodine value of 40) was greater than that of the single oils (SAP value of 90 and an iodine value of 40).
As a result, soap derived from these two oils has unique attributes.
It’s appropriate for screening effective soaps and is superior to single-oil soaps (53).
This research aims to demonstrate the capacity of extra-ordinary oils to be converted into soap. Palm oil is one of the most commonly consumed edible oils. It was employed in the experiment.
Palm oil was repeatedly fried five times, the total polar substances (TPS) content of which increased by 10%, 15%, 20%, and 24%, respectively.
Oil samples were gathered and Soaps were created.
We studied the pH, water content, total alkalinity, and total fat content of the towels.
The amount of malondialdehyde (MDA) was also calculated. The MDA value is one of the primary characteristics that differentiate soap derived from fresh palm oil from that derived from re-fried palm oil (54).
This investigation sought to determine if residual oil from frying can be used to create homemade soap.
This investigation utilized frying oil and unrefined rapeseed oil, sunflower oil, and palm oil (all of which had a total of 24% polar chemicals in their production of handcrafted soap).
Physical, chemical, and microbiological analyses were performed on the samples that were intended to authenticate their authenticity.
Compared to the sunflower oil soap, soaps that had a lower percentage of palm oil and rapeseed oil had a higher degree of harshness.
Sunflower oil soap had the greatest concentration of MDA, it was 6.61 µg/g.
Other oils that had a high concentration of MDA had a maximum of 0.94 µg/g.
These findings demonstrate that this byproduct can be utilized to create homemade soap (55).
This research investigated the physical and chemical properties of different varieties of homemade soap derived from five different oils that were produced in local communities in Nigeria.
The loss of weight was part of the medical exam.
The quantity of moisture and the capacity of the soaps to bubble was evaluated following the procedure.
In the chemical analysis, various metrics, including the saponification value and the number of free fatty acids, were documented. Conventional analytical methods were employed.
The investigation revealed that the oils used to create the samples… many soaps met the criteria for commercial production of bar soap (56).
This investigation sought to assess the quality of solid, obvious soaps and determine the composition of Kananga soaps derived from different types of oils. Moisture content, the volume of free alkali present, pH, hardness, consistency of the foam, and the antibacterial properties of the foam were assessed.
The investigation revealed that the optimal composition of soap had a concentration of 1.5% Kananga oil and coconut oil (VCO). The moisture content was one of the most significant attributes of solid soaps (4.39-1.81%), pH = 11.33-11.81, and the stability of their foam (69.70-85.45%), the hardness of their soap (0.042-0.065 mm/g/s), and the content of free alkali (0.63-0.96%). However, additional research is necessary to increase the ylang-ylang oil content and decrease the amount of free alkali in soap (57).
Three natural soaps were derived via the cold process: a rosehip soap with hibiscus flowers (H), as well as a base/controlled soap (BB).
Forest Woodland (FC) Soap
Based on the component analysis principle (PCA), the unsaponifiable fatty acids in the hibiscus soap were different than those in the other soaps.
These findings show that fatty acid soaps derived from the cold process have a significant impact on the quality, overall experience of taste, and preferences of non-vegetarian soaps (58).
This investigation involved five different oils: coconut oil, palm oil, castor oil, olive oil, and ghee. These oils were instrumental in creating different soaps. These oils were incorporated into different quantities.
This investigation determined the total fat content, mineral content, pH, chemical components that are evaporable, and the moisture content of the towels.
The results were juxtaposed with various popular shows that are commercially viable.
The results showed that the soap derived from olives had a higher pH, a higher total fat content (TFM), and several other beneficial properties in comparison to other soaps. Its pH was 59. Other than the scent, the utilization of small quantities (in domestic settings) has a minimal impact on the quality of the oils. As such, the resulting soap is typically similar to soap derived from fresh oil, but it is more susceptible.
However, over the long haul, the characteristics of the oils have significant changes. This is made up of a darker color, a different scent, and an increase in free fatty acids (FFA) to approximately 5. Because of the higher concentration of essential fatty acids, the soap produced in this way is darker in color and produces less lather than the soap made from fresh vegetable oils (lather grades 14 to 5).
These findings indicate that high quality soaps are possible purchases at a low cost using either light or heavy vegetable oils (60).
Conclusions:
After studying the existing research and the associated topics, we reach the following conclusions:
1. Studies have demonstrated that soap can be derived from various oils, both animal and vegetable-based (10.20).
2. The harshness, scent, and cleaning abilities of soap, as well as the distribution of unsaturated and saturated fatty acids, are all attributes that are desired in soap.
The machine’s capacity to produce moisture and foam (57).
3. Some investigations have examined the utilization of different detergents in the manufacturing of soap, while others have studied the effects of oils on the final properties of soap.
Recommendations:
1. Studies on the use of animal fats in soap making.
2. Studies on the use of vegetable oils in soap making.
3. Studies on the effect of different types of oils on the physical and chemical properties of soap.
Propositions:
After analyzing previous studies’ results, he proposed the following options:
1. Soap is derived from wasted food.
2. Investigate the physical and chemical properties of food waste-based soap.
Reference:
- Abitogun, A.S., Alademeyin, O.J. & Oloye, D.A. (2009). Extraction and characterization of castor seed oil. Internet Journal of Nutrition and Wellness, 8(2).
- Acquaye, D., Smith, M., Letchamo, W., Angers, P. & Simon, J. (2001). Shea butter: Agrobusiness in sustainable natural African plant products. Available at: http://www.aspenwoodbathhandbody.com/Docs/sheabutter.pdf (Accessed: 1 June 2009).
- Adel, Y. Girgis (1999). Using of mucilage palm oil in the toilet soap production. Grasas y Aceites, 50(3), pp. 185–192.
- Ainie, K., Hamirin, K. & Peang-Kean, L. (1996). Chemical and physical characteristics of soap made from distilled fatty acids of palm oil and palm kernel oil. Journal of the American Oil Chemists’ Society, 73, pp. 105–108.
- Aiwize, E.A. & Achebo, J.I. (2012). Liquid soap production with blends of rubber seed oil (RSO) and palm kernel oil (PKO) with locally sourced caustic potash (KOH). Nigerian Journal of Technology, 31(1), pp. 63–67.
- Alty, L.S. (2009). Journal of Chemical Education, 86(8), p. 962.
- Antonić, B. et al. (2020). Physicochemical characterization of home-made soap from waste-used frying oils. Processes. Available at: www.mdpi.com/journal/processes.
- Arasaretnam, S. & Venujah, K. (2019). Preparation of soaps by using different oil and analyze their properties. Natural Products Chemistry & Research, 7(1).
- Batnasan, Z., Natsagdorj, P. & Tserendorj, T. (2022). Preparation of liquid soap from sheep’s tail fat. Open Journal of Applied Sciences, 12, pp. 662–671.
- Berneckė, S. & Maruška, A. (2013). Analysis of free fatty acids in soap samples by means of gas chromatography–mass spectrometry. Department of Biochemistry and Biotechnologies, Vytautas Magnus University, Lithuania, 24(4), pp. 307–311.
- Boadu, K.O., Anang, M.A. & Kyei, S.K. (2017). Chemical characterization of shea butter oil soap (Butyrospermum Parkii). International Journal of Development and Sustainability, 6(10), pp. 1282–1292.
- Dowd, M.K. (1996). Journal of the American Oil Chemists’ Society, 73, p. 1287.
- Dowd, M.K. (1998). Journal of Chromatography A, 816, p. 185.
- Ekop, S.A., Etuk, B.A. & Eddy, N.O. (2007). Effect of some local additives on the chemical constituent of palm oil. Journal of Applied Sciences and Environmental Management, 11(1), pp. 85–89.
- Ekpa, O.D., Akpanabiatu, M., Mauro, A. & Renzo, R. (2000). Differences and polymorphism in palm oil blended with coconut oil. Global Journal of Pure & Applied Science, 2, pp. 277–282.
- Ellis, M. (2008). Colonial soap making: Its history and techniques. Available at: http://www.akasoft.com/soapfat/historycontent.html (Accessed: 7 June 2008).
- Felix, S., Araujo, J., Pires, A.M. & Sousa, A.C. (2017). Soap production: A green perspective. Waste Management, 66, pp. 190–195.
- Ferris, R.S.B., Collinson, C., Wanda, K., Jagwe, J. & Wright, P. (2001). Evaluating the market opportunities for shea nut and shea nut processed products in Uganda. Natural Resources Institute, USAID.
- Firempong, C.K. & Mak-Mensah, E.E. (2011). Chemical characteristics of toilet soap prepared from neem seed oil. Asian Journal of Plant Science and Research, 1(4), pp. 1–7.
- Fintrac Inc. (1999). Market and technical survey: Shea nuts. Washington DC.
- Gandhi, A.P. & Taimini, V. (2009). Organoleptic and nutritional assessment of sesame biscuits. Asian Journal of Food and Agro-Industries, 2(2), pp. 87–92.
- Girish, K. & Shankara, B.S. (2008). Neem – A green treasure. Electronic Journal of Biology, 4(3), pp. 102–111.
- Gunstone, F.D. (2005). Vegetable oils. In: Shahidi, F. (ed.) Bailey’s Industrial Oil and Fat Products. 6th ed. John Wiley & Sons, pp. 224–225.
- Karl, I.Z. (1991). By-product utilization. In: Introduction to fats and oils technology. American Oil Chemists’ Society, pp. 263.
- Khadayesh, H. (2019). Preparing carrot soap (carotene) in two ways, hot and cold. Master’s thesis, University of Kasdi-Merbah Ouargla.
- Maotsela, T., Danha, G. & Muzenda, E. (2019). Utilization of waste cooking oil and tallow for toilet soap production. Procedia Manufacturing, 35, pp. 541–545.
- McGraw-Hill (2007). Encyclopedia of Science and Technology. 10th ed. New York: McGraw-Hill, Vol. 16, pp. 586–587.
- Mohammed, M.I. & Hamza, Z.U. (2008). Physicochemical properties of oil extracts from sesame seeds. Journal of Applied Sciences and Environmental Management, 12(2), pp. 99–101.
- National Research Council (1992). Neem: A tree for solving global problems. Washington, DC: National Academy Press.
- Openshaw, K. (2000). A review of Jatropha curcas. Biomass and Bioenergy, 19, pp. 1–15.
- Phanseil, O.N., Dueno, E. & Xianghong, W.Q. (1998). Synthesis of exotic soaps in the chemistry laboratory. Journal of Chemical Education, 75(5), p. 612.
- Robertson, D.L. (2006). Saponification and making soap. Chemistry 102 Experiments Index. Available at: http://www.miracosta.cc.ca.us/home/dlr/102exp8.htm.
- Spitz, L. (2018). Soap manufacturing technology. 2nd ed. London: Academic Press & AOCS Press.
- Warra, A.A. (2013). A report on soap making in Nigeria using indigenous technology. Kebbi State University of Science and Technology.
- Wolf, R., Tuzun, B. & Tuzun, Y. (2001). Soaps, shampoos and detergents. Clinics in Dermatology, 19, pp. 393–397.

